Master Cell / Microbial Banking
Our cGMP-compliant cell bank can preserve a large volume of cell lines, microbial cells, and other biologics for the manufacture of commercially available therapeutics.
We ensure that all samples collected either on the field or on our biobank are carefully processed and transported using the best techniques for preservation.
Clinical Trial Material
We take part on several preclinical to clinical testing to determine efficacy of medical drugs and substances.
Our dedicated scientist can serve as your partner from lead optimization to commercialization for your next advanced therapy.

cGMP Process Development
Apart from process equipment and turnkey facilities, we offer process development to support client's specific needs and requirements for cell culture process development and optimization. This includes cell culture medium optimization, seed preparation process development, and system layout and process integration.

MSAT (Manufacturing Sciences & Technology)
We are able to provide fast, efficient and robust vaccine technology transfer with single-use solutions, predictive process control, process scale up/scale down, complexity reduction, and process validation.

Cellular Agriculture/Complementary Proteins
We provide clients with end to end contract development and manufacturing services for food ingredient and food product.

Plasmid DNA Manufacturing Services
We offer CDMO services to manufacture various grades of plasmid DNA for preclinical, clinical and commercial production. We maintain GMP standards from the starting material to the final drug products in our Plasmid DNA production process.
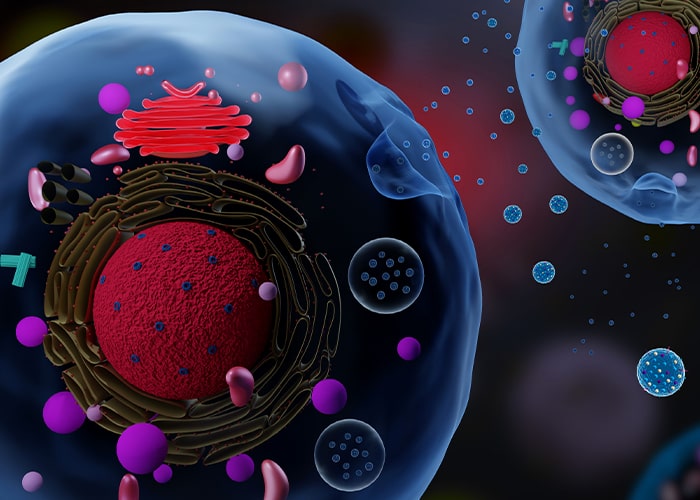
Extracellular Vesicles
Extracellular vesicles are secreted by many cells, of which the common ones are Mesenchymal Stromal Cells, Pericytes, CAR-T cells, human primary bronchial epithelial cell, epithelial cells, EBV(Epstein-Barr virus)-immortalized B cells, HUVECs (Human umbilical vein endothelial cells) Leukocytes, platelets, cancer cells and even neural cells. They are classified into three groups depending on their size.

Cell Line Creation
We specialize and are best-in-class in accelerated adherent cell line creation and development in conjunction with our adherent cell banking, characterization, testing and storage services for your various applications.
Genetically-modified Cell Therapy
At Esco Aster, we leverage our expertise into the production of genetically-modified cell therapy such as CAR-Immune cells (T cells, NK cells, Dendritic cells). Our adaptable and dynamic approaches employed across multiple cell types coupled with cancer experts enable us to provide process design and developmental solutions all the way through GMP manufacture and supply.
Exosome Nasal Formulation Development
Esco Aster offers advanced formulation
solutions for inhalation-based exosome delivery, enabling developers to
transform exosome candidates into nasal spray, nebulizer-ready, or dry powder
inhalation products. By integrating specialized particle engineering, stability
studies, and aerosolization expertise, we support the development of inhaled
exosome therapeutics designed for respiratory, neurological, and systemic
indications.

Drug Product: Aseptic Fill & Finish
Esco Aster
is the first in Southeast Asia (ASEAN) to provide small-batch aseptic
fill-and-finish services post-sterile filtration for further manufacturing,
final drug products and for terminal sterilisation with in house autoclave that
are fully compliant with USP <1207> container-closure integrity testing.

Esco Aster Strategic Capital (Fundraising ) As A Service With Accredited Investors
Esco Aster provides strategic capital support as a value-added fundraising service designed to align capital, capability, and execution. Our approach supports a broad range of stakeholders across the innovation and investment ecosystem, including founders and startups, shareholders, limited and general partners, funds of funds, sovereign funds, institutional and accredited investors, government research bodies and grantors, accelerators, incubators, venture studios, venture capital and private equity firms, foundations, and philanthropists.

Consultancy
Esco Aster is a specialized
biomanufacturing platform with a proven track record of working with startups,
well-funded biotechs, medical and healthcare centers to help them achieve
critical fundraising and investment milestones.

Clinical Development
Esco Aster is a vertically integrated
bioinnovation accelerator delivering end-to-end “idea-to-exit” solutions that
seamlessly bridge preclinical discovery to clinical development with our
concept-to-product CRDMO service offering.
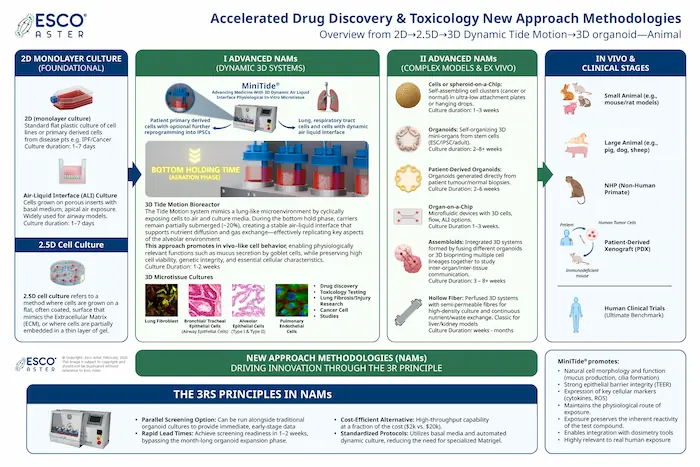
Drug Discovery
At Esco Aster, we believe the best scientific outcomes are achieved through a diverse, complementary toolkit. MiniTide® is offered not as a replacement, but as a high-performance parallel solution that runs alongside traditional 2D and organoid cultures. This approach delivers rapid, high-fidelity data, accelerates decision-making, and significantly de-risks the path to clinical trials.