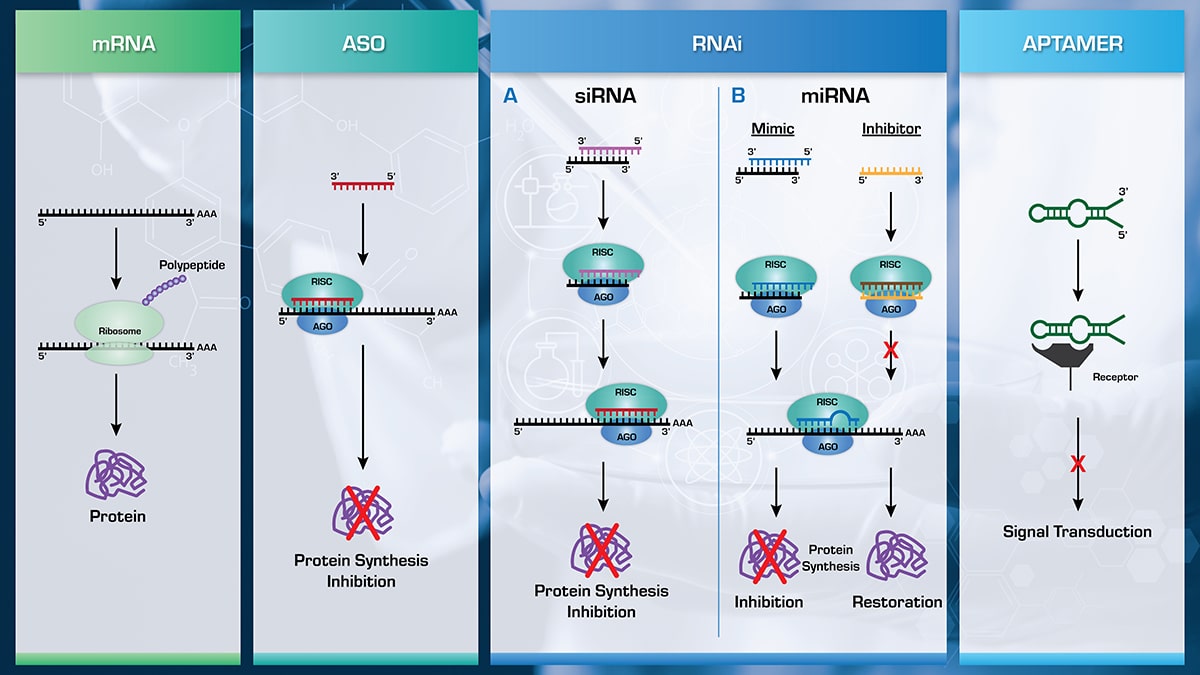
Nanomedicine and mRNA Vaccines
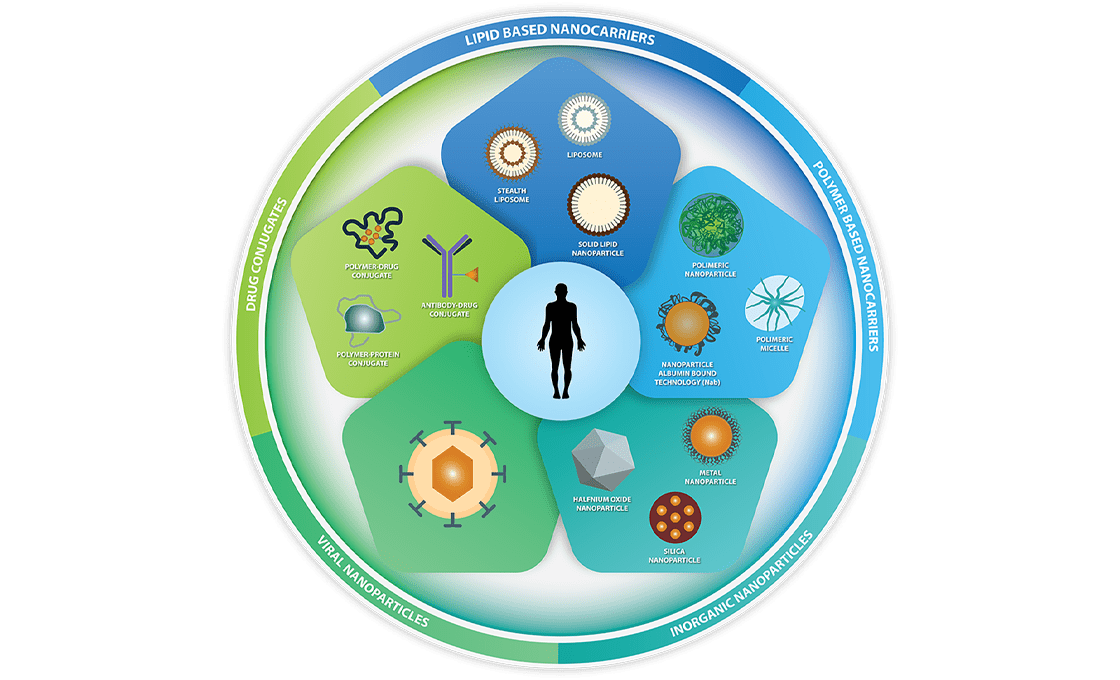
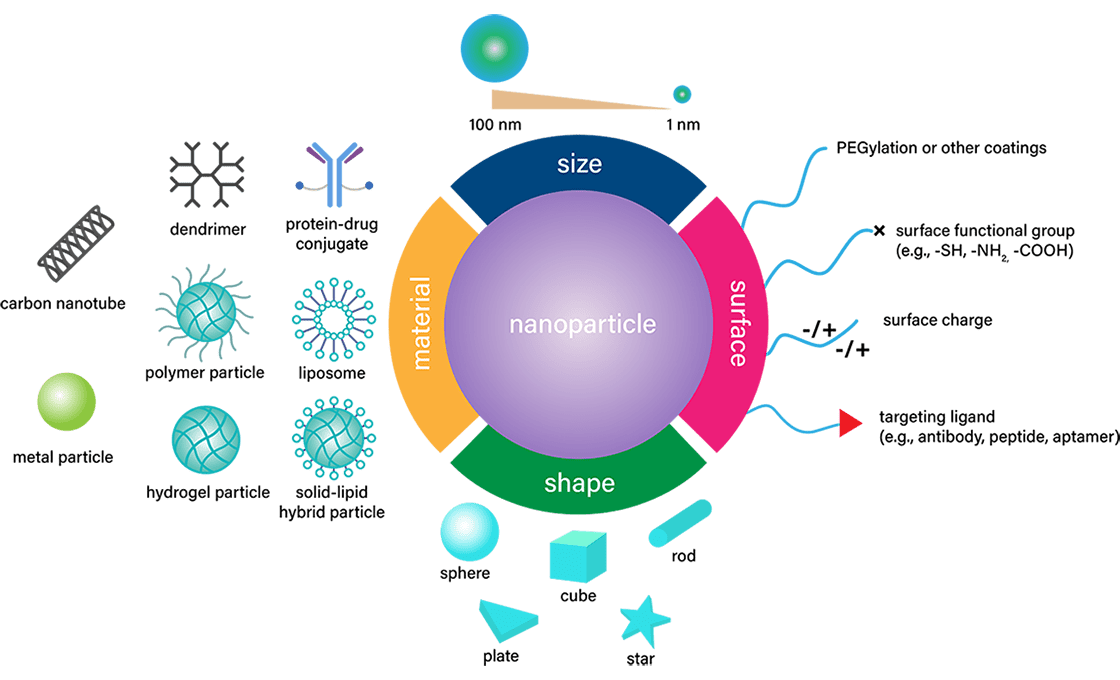
Nanomedicine is ideally placed to upend current standards in disease diagnosis, prevention, and treatment. It is a versatile technology that uses nanoparticle encapsulation of a wide range of substances to achieve a variety of clinical outcomes.
In the area of prevention, nanomedicine has proven to be a useful platform for the delivery of new vaccine modalities. Notably, two of the most widely used COVID-19 vaccines contain mRNA encapsulated in lipid nanoparticles (LNPs). This delivery system improves the stability, shelf life, and efficiency of mRNA delivery within the body.
Nanomedicine in Diagnostics and
Medical Imaging
Nanomedicine has diagnostic applications as well. Various imaging modalities can be delivered using various nanoparticle types. Nanoparticles, for example, have been used to deliver and localize fluorescence markers in fluorescence imaging. The nanoparticles could be further modified to target specific cell types or organs.
Nanomedicine in Peptide and
Nucleic Acid-based Therapeutics
Nanomedicine has the most diverse range of applications in disease treatment.
Encapsulating small molecule drugs, especially those with poor permeability and solubility, within nanoparticles enables their application by overcoming their pharmacokinetic limitations. Larger molecules, such as natural product analogs or their derivatives, can also be successfully delivered with nanoparticles.
Perhaps the most significant benefit of nanoparticle technology would be the ability to develop peptide and nucleic acid-based therapeutics. Because of their poor stability and susceptibility to degradation, peptide and nucleic acid therapeutics have been used sparingly in the past. Liposome encapsulation not only protects the contents from degradation, but in some cases, such as LNPs, it also facilitates cellular delivery of the payloads.
In addition, derivatization of the nanoparticles is another tool that is used to improve the delivery to the target sites, particularly for radioactive or chemotherapeutic agents.