Advancing cGMP-Compliant Manufacturing of Human iPSCs for Clinical Applications

With the conditional approvals in Japan in beginning of 2026 for ipscs for parkinsons disease (Sumitomo) and cardiomycote sheet for severe heart failure (Cuorips) it is now not a question of "if stem cell therapy works" but now the challenge is on biomanufacturing and scaling up cGMP stem cells.
A newly published review by our team highlights significant progress in the development of cGMP-compliant, large-scale manufacturing processes for human induced pluripotent stem cells (iPSCs) including 3D Adherent Tide Motion bioreactors, marking an important step forward in advancing regenerative medicine and cell-based therapies.
Human iPSCs have emerged as a transformative platform in modern biomedical research, offering vast potential in disease modelling, drug discovery, and regenerative medicine. Their ability to differentiate into various cell types makes them highly valuable for therapeutic applications. However, a key challenge in translating iPSC technologies from research to clinical use lies in establishing robust, scalable, and regulatory-compliant manufacturing systems that ensure consistent quality and safety.
The review addresses these challenges by presenting advancements in large-scale iPSC manufacturing under current Good Manufacturing Practice (cGMP) conditions. The work outlines strategies to improve scalability, reproducibility, and process control, all of which are essential for producing clinical-grade cell therapy products.
Key highlights from the study include:
- Implementation of cGMP-compliant workflows to support clinical translation
- Development of scalable expansion strategies for iPSC production
- Emphasis on process consistency and quality control to meet regulatory requirements
- Strengthening the foundation for safe and effective therapeutic applications
In alignment with these advancements, Esco Aster continues to support the evolution of scalable cell therapy manufacturing through its innovative Tide Motion technology. Designed to enhance cell expansion and process consistency, Tide Motion enables more efficient and controlled culture environments, addressing key bottlenecks in large-scale iPSC production. This technology supports improved yield, reproducibility, and scalability, making it a valuable solution for translating iPSC-based therapies into clinical and commercial settings.
These developments are critical in bridging the gap between early-stage research and real-world clinical deployment. By enabling reliable and standardized manufacturing processes, this work—alongside enabling technologies such as Tide Motion—supports the broader adoption of iPSC-based therapies across a range of indications.
As the field of regenerative medicine continues to evolve, the establishment of scalable and compliant manufacturing platforms will be central to unlocking the full therapeutic potential of stem cell technologies. This publication contributes to the growing body of knowledge driving innovation in biomanufacturing and reinforces the importance of integrating quality, scalability, and regulatory readiness into cell therapy development.
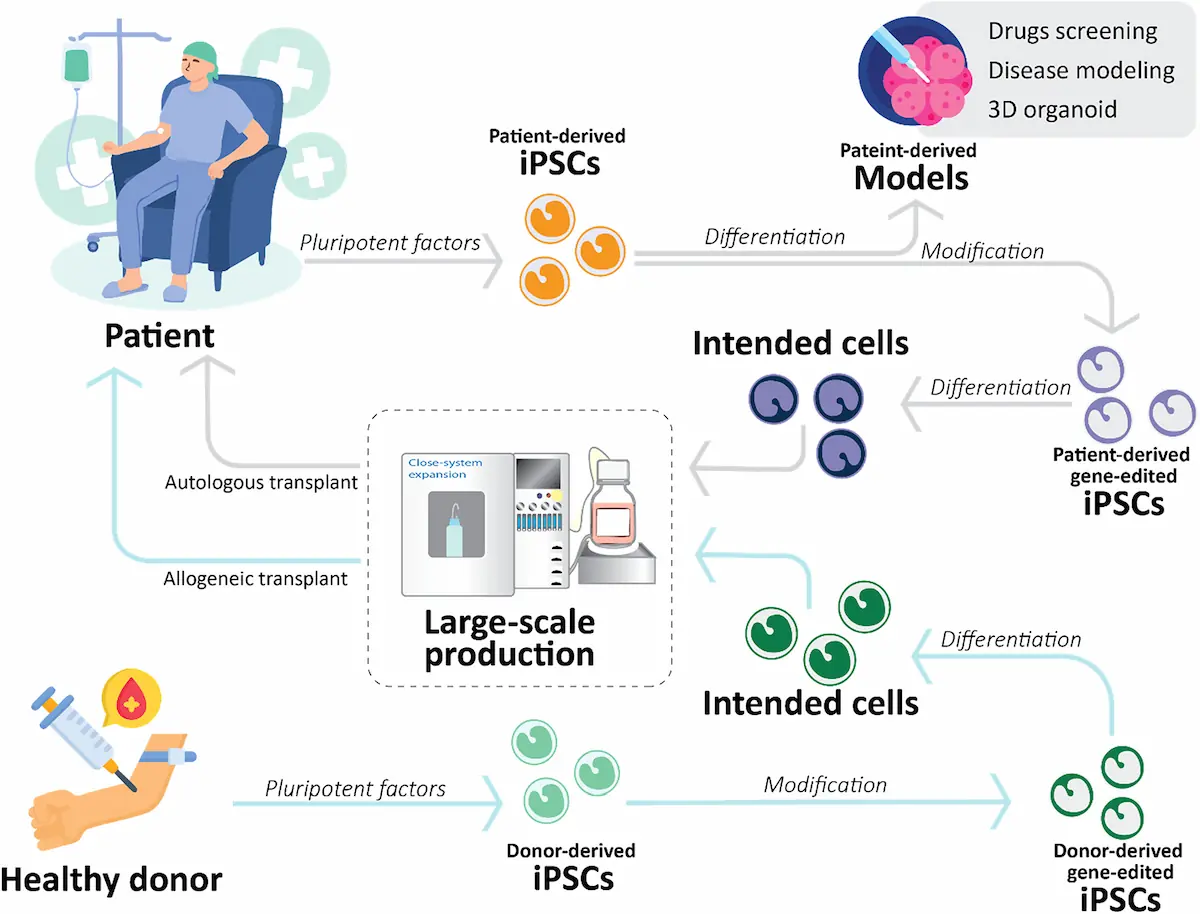
Ipsc-Based Disease Modeling And Therapeutic Strategies
For more information, access the full publication here:
https://www.sciencedirect.com
https://www.isct-cytotherapy.org
About Esco Aster
Esco Aster is a leading provider of cell therapy and biomanufacturing solutions, focused on enabling the scalable production of advanced therapies. Through innovative technologies and integrated manufacturing capabilities, Esco Aster supports partners in translating scientific breakthroughs into clinically viable treatments.
Collaboration Opportunities
We welcome opportunities to collaborate with academic institutions, biotech companies, and clinical partners to advance iPSC-based therapies and scalable manufacturing solutions. If you are interested in partnering with us, please contact us at [email protected] to explore potential collaborations.